Over, Herbert
Surface Chemistry and Model Catalysis
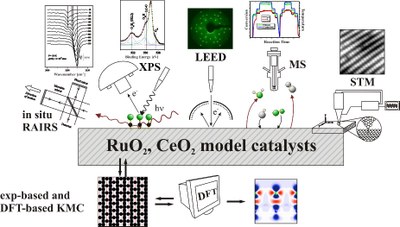
In surface electrocatalysis, we develop and characterize novel ultrathin single crystalline metallic oxide electrodes, which are employed in the kinetic and stability studies of oxygen and chlorine evolution reaction. One of the major objectives is to unravel the elementary reaction steps in these evolution reactions and constructing the free energy profile using both electrochemical techniques and theoretical ab-initio methods. We conduct synchrotron based experiments to study in-situ the corrosion processes of such model electrodes under strongly oxidizing conditions.
Contact
Prof. Dr. Herbert Over
Institute of Physical Chemistry
Phone: +49-641-99-34550
Fax: +49-641-99-34599
Chemistry building, Heinrich-Buff-Ring 17, Room B 41

Applications/Functionalities:
- (Photo-)Electrocatalysis
- Catalysis
- Surface Technologies
Methods:
- Chemical Analytics
- Electrochemical Measurements
- Mass Spectrometry
- Scanning Probe Methods
- Vibrational Spectroscopy
- Structural Analytics
Classes of Materials:
- Thin Films
- Solid Electrolytes / mixed Conductors
- Metal Anodes
- Nanomaterials
- Oxides
